Unlocking the Secrets of Electron Affinity Trends: What Affects It?
Electron affinity is a fundamental concept in chemistry that helps us understand the behavior of atoms and molecules. It refers to the energy change that occurs when an electron is added to a neutral atom in its gaseous state. This concept is crucial in understanding the periodic trends in the periodic table and has significant implications in fields such as materials science, chemistry, and physics. But what affects electron affinity trends? In this article, we will delve into the world of electron affinity and explore the factors that influence it.
As chemist and Nobel laureate Linus Pauling once said, "The laws of thermodynamics govern the behavior of atoms and molecules, and the electron affinity is a fundamental aspect of these laws." Understanding electron affinity trends is essential in predicting the chemical behavior of elements and compounds. However, determining the electron affinity of an element can be challenging due to the complexity of the processes involved. The electron affinity of an element is affected by various factors, including atomic size, electronegativity, and electron configuration.
Atomic Size: A Key Factor in Electron Affinity Trends
One of the primary factors that affect electron affinity is atomic size. As we move down a group in the periodic table, the atomic size increases due to the addition of new energy levels. This increase in atomic size results in a decrease in the effective nuclear charge, making it easier for an electron to be added to the atom. Consequently, the electron affinity becomes more negative, indicating a greater tendency to attract an electron.
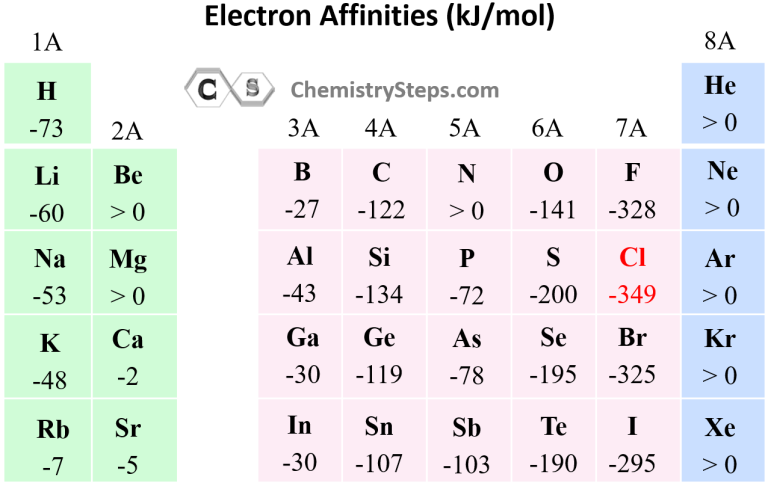
To illustrate this trend, let's consider the electron affinity of the halogens in Group 17. The electron affinity of fluorine (F) is -328 kJ/mol, while that of iodine (I) is -295 kJ/mol. As we move down the group, the atomic size increases, resulting in a decrease in the effective nuclear charge and a corresponding decrease in the electron affinity. This trend is consistent across the periodic table, with elements having larger atomic sizes exhibiting lower electron affinities.
Electronegativity: A Measure of Attraction to Electrons
Electronegativity is another critical factor that affects electron affinity trends. Electronegativity is a measure of an atom's ability to attract and hold onto electrons. The more electronegative an atom is, the more it tends to attract electrons. Consequently, the electron affinity of an atom is influenced by its electronegativity.
As chemist and Nobel laureate William Hodgson Krogmann once said, "Electronegativity is a measure of the ability of an atom to attract and hold onto electrons. The higher the electronegativity, the greater the tendency to attract electrons." To illustrate this trend, let's consider the electron affinity of the chalcogens in Group 16. The electron affinity of oxygen (O) is -141 kJ/mol, while that of sulfur (S) is -200 kJ/mol. As we move down the group, the electronegativity decreases, resulting in a decrease in the electron affinity.
Electron Configuration: A Critical Factor in Electron Affinity Trends
Electron configuration is another important factor that affects electron affinity trends. The electron configuration of an atom determines the arrangement of electrons in its energy levels. The more stable the electron configuration, the less likely an atom is to attract an electron. Conversely, if the electron configuration is unstable, the atom is more likely to attract an electron, resulting in a more positive electron affinity.
To illustrate this trend, let's consider the electron affinity of the transition metals in the d-block. The electron affinity of chromium (Cr) is -139 kJ/mol, while that of manganese (Mn) is -232 kJ/mol. As we move across the d-block, the electron configuration becomes more stable, resulting in a decrease in the electron affinity. This trend is consistent across the periodic table, with elements having more stable electron configurations exhibiting lower electron affinities.
Exceptions to the Trends: A Closer Look
While the trends discussed above provide a general understanding of electron affinity trends, there are exceptions to these trends. Some elements exhibit electron affinities that contradict the expected trends. For example, the electron affinity of carbon (C) is +117 kJ/mol, while that of nitrogen (N) is -172 kJ/mol. This discrepancy can be attributed to the fact that carbon has a unique electron configuration, which results in a higher electron affinity.
Another exception to the trend is the electron affinity of fluorine (F). Despite being a highly electronegative atom, fluorine exhibits a relatively low electron affinity due to its small atomic size and high ionization energy.
Conclusion
In conclusion, electron affinity trends are influenced by various factors, including atomic size, electronegativity, and electron configuration. Understanding these trends is essential in predicting the chemical behavior of elements and compounds. While there are exceptions to these trends, the general principles discussed above provide a comprehensive understanding of electron affinity trends. As chemist Linus Pauling once said, "The laws of thermodynamics govern the behavior of atoms and molecules, and the electron affinity is a fundamental aspect of these laws."
By understanding the factors that affect electron affinity trends, we can gain a deeper insight into the behavior of atoms and molecules. This knowledge has significant implications in fields such as materials science, chemistry, and physics, and has the potential to lead to new discoveries and innovations.